 To add the C4 term, see Tutorial 2.4 in the 12-6-4 nonbonded section. The parameters listed below contain an added r -4 term and reproduce the experimental hydration free energy (HFE), ion-oxygen distand (IOD) and coordination number (CN) values without significant compromise. These ion parameters were parameterized against the experimental hydration free energies and/or ion-oxygen distances. Li +,Na +, K +,Rb +,Cs +,Tl +,Cu +,Ag +,NH 4 +,H 3O +, F -,Cl -,Br -,I -ĭivalent, trivalent and tetravalent also available - see manual 
Load the ions lib file and matching water-model frcmod file: This consistent set of parameters for alkali halide ions were developed by fitting solvationįree energies, radial distribution functions, ion-water interaction energies and crystal lattice energies and latticeĬonstants for non-polarizable spherical ions. Ion Parameter Set Availability and Implementation Joung and Cheatham 1 To use the 12-6-4 model see Tutorial 2.4 in the 12-6-4 nonbonded section. One needs to source the leaprc file for the water and then load the 12-6-4 frcmod file by itself, and then use ParmEd to add the C4 terms. To use the 12-6-4 parameters requires an extra step to insure the C4 term is implemented. 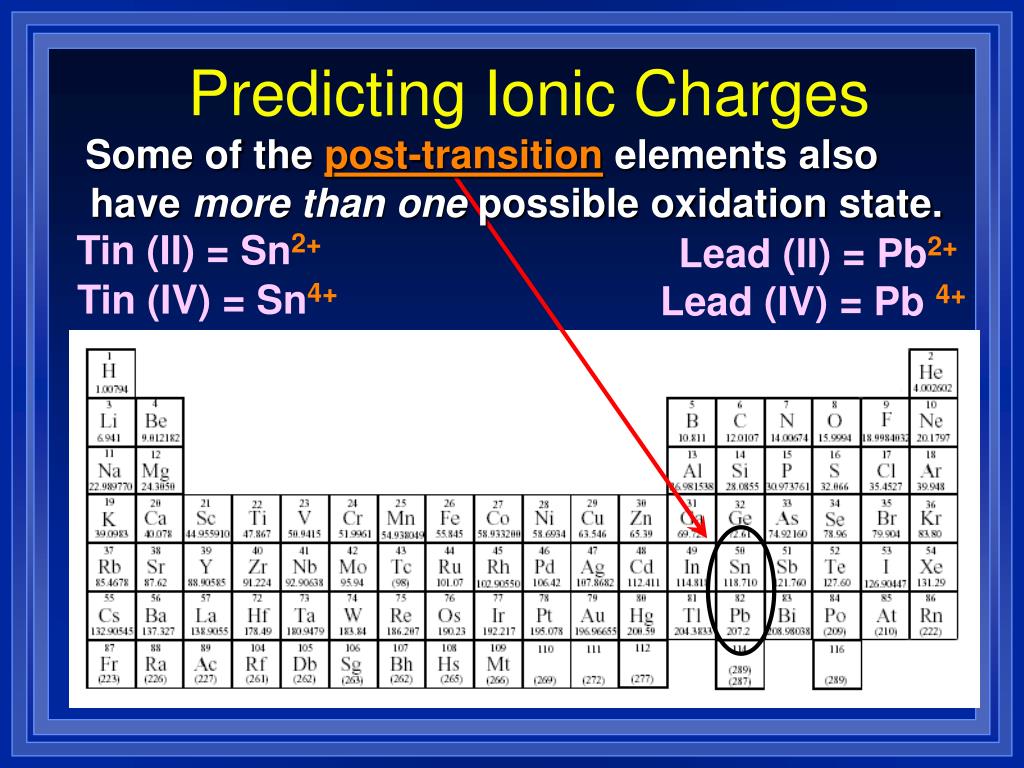
However ion parameters are chosen, they should be consistent with each other (all 12-6 or all 12-6-4) and the water model.įor highly charged metal ions, the 12-6-4 LJ-type nonbonded model performs much better than the You should read each article and understand the pros and cons of each. 
Some ions are parameterized against hydration free energies (experimental or TI calculations), ion-oxygen distances and/or lattice energies. Ideally the ions are chosen to be compatible with the water model. ) will require a set of parameters (lib and frcmod file). Just like each type of biomolecule, each ion (Na +, K +, Mg 2+, Cl -, etc. Several other pairs of radical cation/even electron adenine–silver complexes were also found to undergo different fragmentation reactions.For a full list and explanation of different ion parameters,Ĭhoosing parameters for ions can be tricky. radical cation, which in turn fragments quite differently to the even electron + ion. It undergoes a silver atom loss to form the +. −H] + ion adds two neutral reagents in ion–molecule reactions, consistent with the presence of two vacant coordination sites. + Ag 2] 2+ ions suggest that planar centrosymmetric cations, in which two adjacent silver atoms are bridged by two N7H adenine tautomers via N(3),N(9)-bidentate interactions, are the most stable. DFT calculations on several isomeric [Ad 2 − H] + ion and does not undergo proton transfer to butylamine. − H] + fragments via loss of neutral adenine to form the [Ad + Ag 2 + Ag 2] 2+ is observed to dissociate to + and to react with butylamine via proton transfer, while [Ad 2 
Z)+ complexes play pivotal roles in directing the types of fragmentation and ion–molecule reactions observed. Both the charge and the stoichiometry of the [Ad x We find that the most stable + ion does not correspond to the most stable neutral adenine tautomer, consistent with previous studies that have shown that transition metal ions can stabilize rare tautomeric forms of nucleobases. DFT calculations of neutral adenine tautomers and their silver ion adducts provide insights into the binding modes of adenine. Density functional theory (DFT) calculations have been used to examine potential structures for several of the smaller ions while multistage mass spectrometry experiments have been used to probe their unimolecular reactivity ( via collision-induced dissociation (CID)) and bimolecular reactivity ( via ion–molecule reactions with the neutral reagents acetonitrile, methanol, butylamine and pyridine). Electrospray ionization (ESI) of solutions containing adenine and AgNO 3 yields polymeric [Ad x 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
